Stryker InSpace Balloonplasty
What is InSpace Biodegradable Implantable Balloon?
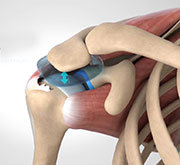
InSpace Biodegradable Implantable Balloon is a new treatment option for the treatment of massive irreparable rotator cuff tears of the shoulder. It involves implantation of a biodegradable balloon spacer called InSpace Balloon or ISB into the subacromial space of the shoulder to help heal ruptured or torn rotator cuff muscles.
InSpace Balloon, also called Orthospace balloon, acts as a substitute for the fluid-filled sac called bursa (that protects and cushions your shoulder joints) that usually becomes inflamed and painful when the rotator cuff muscles rupture. The Ortho-Space creates adequate room between the shoulder and arm’s scapula, acromion, and humerus bones thereby providing pain-free and friction-free shoulder motion.
The InSpace biodegradable implantable balloon (spacer) is utilized as a subacromial spacer to minimize friction between the acromion and the rotator cuff or humeral head to enable smooth movement of the humeral head against the acromion. The balloon may be placed arthroscopically or through a mini-open procedure. Early biomechanical studies have indicated that Stryker balloon effectively restores normal humeral head position and glenohumeral joint mechanics with improvements in shoulder range of motion, pain, and overall function.
Anatomy of the Shoulder
The shoulder joint is a ball and socket joint with the head of the humerus (upper arm bone) forming the ball, and the cup-shaped depression on the shoulder blade (glenoid fossa) forming the socket. The rotator cuff is a group of four tendons that join the head of the humerus to the deeper shoulder muscles to provide stability and mobility to the shoulder joint. Major injury to these tendons may result in a tear of these tendons and the condition is called rotator cuff tear. The tear causes severe pain, weakness of the arm, and a crackling sensation on moving the shoulder in certain positions. There may be stiffness, swelling, loss of movement, and tenderness in the front of the shoulder.
The subacromial space is the space between the acromion and the top surface of the humeral head. The space houses several soft tissues such as the rotator cuff, bicep tendon, and bursa, which are instrumental in the proper functioning of the shoulder.
Indications InSpace Biodegradable Implantable Balloon
The main indication for an InSpace shoulder spacer is to treat irreparable, massive rotator cuff tears. Your surgeon may recommend placement of a subacromial balloon in the following conditions:
- Elderly patients and those with multiple comorbidities and contraindication to general anesthesia or surgery
- Individuals who are not suitable candidates for standard tendon transfer treatment for rotator cuff repair
- Patients who manifest proximal humeral migration secondary to massive full-thickness rotator cuff tears
- Individuals who have preserved passive range of motion (ROM)
- Elderly individuals suffering from trauma or osteoarthritis-associated degradation
- Failure of conservative treatment such as medications and physical therapy in treating irreparable rotator cuff tears
Preparation for InSpace Biodegradable Implantable Balloon
Preoperative preparation for shoulder spacer surgery may involve the following steps:
- A thorough examination is performed by your doctor to check for any medical issues that need to be addressed prior to surgery.
- Depending on your medical history, social history, and age, you may need to undergo tests such as bloodwork and imaging to screen for any abnormalities that could compromise the safety of the procedure.
- You will be asked if you have allergies to medications, anesthesia, or latex.
- You should inform your doctor of any medications, vitamins, or supplements that you are taking.
- You may need to refrain from supplements or medications such as blood thinners or anti-inflammatories for a week or two prior to surgery.
- You should refrain from alcohol or tobacco at least a few days prior to surgery.
- You should not consume solids or liquids at least 8 hours prior to surgery.
- You should arrange for someone to drive you home after surgery.
- A written consent will be obtained from you after the surgical procedure has been explained in detail.
Procedure for InSpace Biodegradable Implantable Balloon
The InSpace Balloon insertion procedure is similar to other standard rotator cuff operations and is commonly performed in a minimally invasive arthroscopic technique under local anesthesia. The procedure generally involves the following steps:
- After cleaning the skin over the shoulder area, your surgeon makes a few small incisions in your shoulder joint.
- An arthroscope, a slender tubular device attached with a light and a small video camera at the end is inserted through one of the incisions into your shoulder joint.
- The video camera transmits the image of the inside of your shoulder joint onto a television monitor for your surgeon to view.
- Your surgeon then uses small surgical instruments through the other tiny incisions to perform surgical debridement of the joint area (removal of damaged tissue). Care is taken to prevent damage to healthy tissue.
- The Orthospace balloon spacer is implanted between the humeral head and the acromion. The Ortho-Space is made up of a synthetic biodegradable material called poly-L-lactide (PLLA) that gradually breaks down over 12 months and is inflated with a normal saline solution.
- The inflated spacer decreases subacromial friction and creates more space between the humeral head and acromion and coracoacromial arch.
- After adequate repair of the torn rotator cuffs, the balloon decreases friction forces and maintains the structure and function of the repaired cuff.
- The balloon deflates after 2 months and the PLLA gets resorbed completely after 15 months.
Postoperative Care Instructions and Recovery
In general, postoperative care instructions and recovery after shoulder balloon implantation involves the following steps:
- You will be transferred to the recovery area to be monitored until you are awake from the anesthesia.
- Your nurse will monitor your blood oxygen level and other vital signs as you recover.
- You may notice pain, swelling, and discomfort in the shoulder area. Pain and anti-inflammatory medications are provided as needed.
- Antibiotics are also prescribed for risk of infection associated with surgery.
- You may also apply ice packs on the shoulder to help reduce swelling and pain.
- You are advised to walk as frequently as possible to prevent the risk of blood clots.
- Keep your surgical site clean and dry. Instructions on surgical site care and bathing will be provided.
- Refrain from smoking as it can negatively affect the healing process.
- Refrain from strenuous activities and lifting heavy weights for a month or two. Gradual increase in activities over a period of time is recommended.
- An individualized physical therapy protocol is designed to help strengthen your shoulder muscles and optimize shoulder function.
- You will be able to resume your normal activities in a couple of months; however, return to sports may take about 6 months or longer.
- Refrain from driving until you are fully fit and receive your doctor’s consent.
- A periodic follow-up appointment will be scheduled to monitor your progress.
Advantages of InSpace Biodegradable Implantable Balloon
Some of the benefits of subacromial balloon spacer placement include:
- Improved shoulder range of motion
- Safe and low risk of complications
- Minimal postoperative pain
- Minimal damage to muscles
- Quick rehabilitation
- Minimal technical demands
- Shorter operative duration
- Cost-effective
Risks and Complications
Stryker balloon implantation is a relatively safe procedure; however, as with any surgery, some risks and complications may occur, such as:
- Infection
- Bleeding
- Inflammation
- Damage to surrounding soft-tissue structures
- Blood clots (deep vein thrombosis)
- Implant migration
- Local foreign body response
- Wound irritation